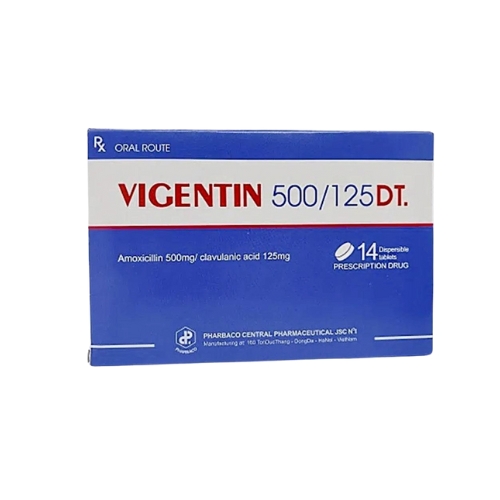
VIGENTIN 500/125 DT
What is Vigentin 500/125 DT Dispersible Tablet?
Vigentin 500/125 DT dispersible tablet is a combination antibiotic containing amoxicillin and clavulanic acid. It is used to treat infections of the respiratory tract, urinary and genital systems, skin and soft tissue, bones and joints, and dental infections.
Composition of Vigentin 500/125 DT Dispersible Tablet
Active Ingredients:
-
Amoxicillin (as amoxicillin trihydrate) 500 mg
-
Clavulanic acid (as potassium clavulanate mixed with microcrystalline cellulose in a 1:1 ratio) 125 mg
Excipients:
- Microcrystalline cellulose, crospovidone, Aerosil R200, magnesium stearate, aspartame, orange flavor, sufficient to make 1 tablet.
Indications
- Upper respiratory infections: Tonsillitis, sinusitis, otitis media not responding to previous treatment
- Lower respiratory infections: Acute and chronic bronchitis, bronchopneumonia
- Urinary and genital infections: Cystitis, urethritis, pyelonephritis
- Skin and soft tissue infections: Boils, abscesses, wound infections
- Bone and joint infections: Osteomyelitis
- Dental infections: Tooth abscess
- Post-abortion infections
How to use Vigentin 500/125 DT dispersible tablets
Dosage
-
Adults and children over 40 kg: 1 tablet (500 mg amoxicillin / 125 mg clavulanic acid) every 12 hours. For severe infections and respiratory infections: 1 tablet every 8 hours.
-
Elderly: No dose adjustment required unless creatinine clearance is < 30 ml/min.
-
Children under 40 kg: Vigentin 500/125 DT dispersible tablets should not be used.
-
Renal impairment:
-
Creatinine clearance > 30 ml/min: No dose adjustment needed.
-
Creatinine clearance 10–30 ml/min: 250–500 mg every 12 hours.
-
Creatinine clearance < 10 ml/min: 250–500 mg every 24 hours.
-
Hemodialysis: 250–500 mg every 24 hours, taken during and after dialysis.
-
Side effects of Vigentin 500/125 DT dispersible tablets
Adverse effects are generally dose-related. When using the combination of amoxicillin and potassium clavulanate, apart from gastrointestinal side effects occurring more frequently, other adverse reactions typically appear with similar frequency and severity as when using amoxicillin alone. Side effects with the oral combination are usually mild and transient, and fewer than 3% of patients need to discontinue treatment.
Common, ADR>1/100
-
Gastrointestinal: Diarrhea (9%), nausea, vomiting (1–5%). Nausea and vomiting are dose-related to clavulanic acid (a 250 mg dose increases risk by 40% compared to a 125 mg dose).
-
Skin: Rash, itching (3%).
Uncommon, 1/1000
-
Blood: Eosinophilia.
-
Liver: Hepatitis and cholestatic jaundice, elevated transaminases. These may be severe and prolonged for several months.
-
Other: Vaginitis due to Candida, headache, fever, fatigue.
Rare, ADR<1/1000
-
Systemic: Anaphylaxis, angioedema.
-
Blood: Mild thrombocytopenia, leukopenia, hemolytic anemia.
-
Gastrointestinal: Pseudomembranous colitis.
-
Skin: Stevens–Johnson syndrome, erythema multiforme, exfoliative dermatitis, toxic epidermal necrolysis.
-
Kidneys: Interstitial nephritis.
Note
Precautions
- Pay attention to elderly patients and those with a history of jaundice or liver dysfunction caused by amoxicillin and potassium clavulanate, as clavulanic acid increases the risk of cholestasis in the liver. Signs and symptoms of cholestatic jaundice are rare when using this medicine but can be severe. However, these symptoms are usually reversible and may resolve within 6 weeks after stopping treatment. Severe hypersensitivity reactions may occur in patients with a history of allergy to penicillin or other allergens; therefore, before starting treatment with amoxicillin, a thorough assessment of any history of allergy to penicillin, cephalosporins, and other allergens is required.
- Patients with moderate or severe renal impairment should have their dosage adjusted (see Dosage section). Amoxicillin users may develop a rash accompanied by fever and swollen lymph nodes.
- Prolonged use of the medicine may sometimes lead to the overgrowth of resistant bacteria.
- Hematologic parameters and liver and kidney function must be periodically monitored throughout treatment. Differential diagnosis is necessary to identify cases of C. difficile–associated diarrhea and pseudomembranous colitis. There is a high risk of rash in patients with infectious mononucleosis.
- Patients with phenylketonuria or those who need to control phenylalanine intake should be cautious when taking tablets containing aspartame, as aspartame is metabolized in the gastrointestinal tract into phenylalanine.
* Use of the medicine in pregnant and breastfeeding women:
- There are not enough data to confirm the safety of the medicine for pregnant or breastfeeding women.
Pregnant women:
- Experimental studies on the reproductive process in rats have shown that oral and injectable administration of the product does not cause malformations. However, due to limited experience with use in pregnant women, the medicine should be avoided during pregnancy, especially in the first trimester, unless necessary as directed by a physician.
Breastfeeding women:
- The medicine may be used during breastfeeding. It does not harm breastfed infants except for the potential risk of sensitization, as a very small amount of the medicine is excreted in breast milk.
* Effects of the medicine on the ability to drive and operate machinery:
- No reports have been found in the reference documents.
Drug Interactions
- The medicine may prolong bleeding and clotting times. Caution is required in patients being treated with anticoagulants (warfarin).
- Like other broad-spectrum antibiotics, the medicine may reduce the effectiveness of oral contraceptives, so patients must be informed in advance.
- Nifedipine increases the absorption of amoxicillin.
- In patients with hyperuricemia, the concomitant use of allopurinol with amoxicillin increases the risk of amoxicillin-induced rash.
- There may be an antagonistic interaction between the bactericidal agent amoxicillin and bacteriostatic agents such as fusidic acid, chloramphenicol, and tetracyclines.
- Amoxicillin reduces the excretion of methotrexate, increasing its gastrointestinal and hematologic toxicity.
- Probenecid taken immediately before or together with amoxicillin reduces the excretion of amoxicillin, thereby increasing its blood concentration. However, probenecid does not affect the half-life, maximum concentration (Cmax), or area under the concentration–time curve (AUC) of clavulanic acid.
Product Information
Storage: Store in a tightly closed container, in a dry place, at temperatures below 30°C.
Packaging: Box of 2 blisters x 7 tablets.
Brand: Pharbaco.
Manufacturer: Central Pharmaceutical Joint Stock Company No. 1 – Pharbaco (Vietnam).